One bad label. That is all it takes. A potency number that does not match the COA, a missing warning line, a health claim buried in the product description. Any of these can trigger an FDA warning letter, a state enforcement pull, or a full product recall that wipes out an entire production run worth of inventory.
CBD brands learned this the hard way in 2024 and 2025. The FDA's CBD regulation page shows a growing list of enforcement actions tied to packaging and labeling violations, not product quality. The irony is brutal. The CBD inside the bottle could be perfectly formulated, third-party tested, and legitimately beneficial. But if the box it ships in does not meet federal and state packaging standards, none of that matters.
Here is what we see working with CBD brands on custom packaging every month: the ones who build compliance into the design phase save thousands. The ones who treat packaging as an afterthought end up redesigning, reprinting, and relabeling under pressure.
Child-Resistant Packaging for CBD Products
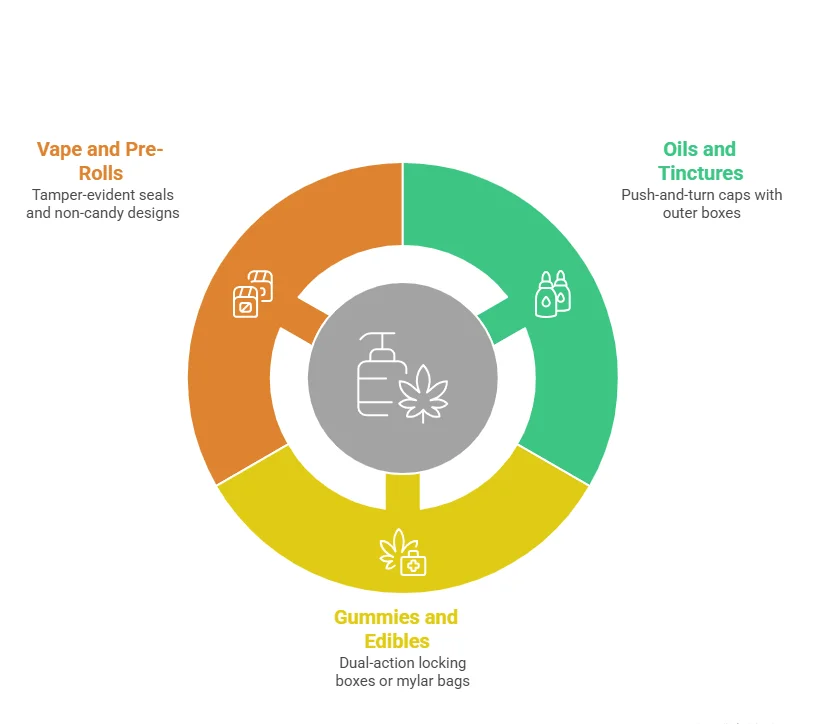
This one is black and white. If the product goes into a person's mouth, the packaging must be child-resistant. Oils, tinctures, gummies, capsules, edibles, dissolvable tablets. All of them. Most states extend this to vape cartridges and concentrates too.
The legal standard comes from the Poison Prevention Packaging Act (PPPA), enforced by the CPSC under 16 CFR §1700.20. And here is where brands get tripped up: "child-resistant" is not a design feature you can eyeball. The packaging system, meaning the specific container paired with the specific closure, must be independently tested and certified. A box that feels tough to open but has never been through protocol testing is not compliant. Regulators do not care how sturdy it looks.
Oils, Tinctures, and Capsules:
Push-and-turn caps on the bottle work for most markets when paired with a tuck end or rigid outer box. But the cap alone does not count unless it has been tested with that exact bottle. Ask the packaging supplier for CR documentation that matches the configuration you are ordering. If they cannot produce it, find a supplier who can.
Gummies, Edibles, and Dissolvables:
Dual-action locking boxes or resealable mylar bags with pinch-and-pull seals handle both child resistance and freshness. Products meant for multiple uses need to re-close with the same CR mechanism every time. A few states, California being the loudest, now want dual-layer protection: a child-resistant inner container inside a child-resistant outer package.
Vape and Pre-Roll Products:
Tamper-evident seals on top of child-resistant closures. That is the expectation in most regulated markets for pre-roll packaging and vape cartridge boxes. And there is a separate rule that catches more brands than any other: the packaging cannot look like it belongs in a candy aisle. No cartoons. No bright, playful color palettes. No design elements that could appeal to anyone under 21. This is actively enforced, and it is the violation that generates the most state-level penalties.
What Has to Be on Every CBD Label
The mandatory elements are specific, and there is no room for creative interpretation.
Product name with a clear CBD or cannabidiol callout. Total CBD in milligrams, both per serving and per package. Full ingredient list in descending order. Manufacturer name and physical address. Batch or lot number tied to lab testing. Net weight or volume. THC declaration if the product contains any detectable amount, which must stay at or below 0.3% delta-9 THC under the 2018 Farm Bill.
A proposed federal rule heading into late 2026 would cap total THC at 0.4mg per finished retail container. If that passes, brands will need to rethink container sizing, serving counts, and how potency panels are structured. Smart brands are already building flexibility into their label templates.
Warning Language That States Require:
"Keep out of reach of children." "For use only by adults 21 and older." "Not intended to diagnose, treat, cure, or prevent any disease." Those three show up in nearly every state. California stacks Prop 65 warnings on top. New York dictates minimum font sizes for mandatory disclosures. Selling in multiple states? Build the label around the strictest state in the distribution map. One universal SKU costs less than reprinting for every new market.
QR Codes and Lab Transparency:
More states now require a scannable QR code on the packaging that links directly to batch-specific Certificate of Analysis (COA) results. Even in states where it is not yet mandatory, printing one is smart. Consumers scan them. Retailers check them. Regulators use them as a first-pass compliance screen. The code needs to link to a live page with actual lab data for that specific batch. A generic homepage link or a dead URL creates a compliance gap and tanks consumer trust in one scan.
Health Claims Will Get a Brand Shut Down:
This is the line most CBD brands struggle with because the temptation is real. The product works. Customers send testimonials. The founder genuinely believes in the benefits. None of that matters on a label.
Words like "cures," "treats," "prevents," "therapeutic," and "medicinal" are drug claims under FDA rules. No CBD product currently has FDA approval to make those claims. Printing them on a box is the single fastest path to a warning letter.
Stick with wellness language. "Formulated for daily wellness." "Supports calm and relaxation." "Made with full-spectrum hemp extract." These phrases communicate the product's positioning without tripping FDA triggers. If the marketing team pushes for stronger language, run it past legal counsel before it hits the printer. Reprinting 5,000 boxes after a warning letter costs a lot more than one legal review.
State Rules That Change the Box Design
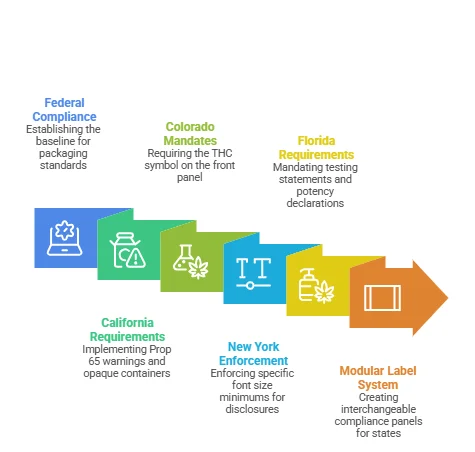
Federal compliance is the floor, not the ceiling. Every state adds its own layer, and the differences show up directly in packaging design decisions.
California wants Prop 65 warnings, opaque containers for edible CBD, and resealable packaging. Colorado mandates the universal THC symbol at a minimum of half an inch in diameter on the front panel. New York enforces specific font size minimums on all disclosures. Florida requires testing statements and potency declarations on every hemp-derived CBD product sold in the state.
For brands selling across three, five, or fifteen states, the most efficient approach is a modular label system. Lock the core brand elements (logo, color palette, product name) and build interchangeable compliance panels for state-specific requirements. It avoids full box redesigns every time a new state gets added to the distribution list.
Making CBD Packaging Look Premium and Legal
Compliance constraints do not kill great design. They focus it. The CBD brands winning shelf space in 2026 treat regulations as guardrails, not roadblocks.
Matte lamination with a foil-stamped logo on a rigid box delivers a luxury feel while leaving plenty of real estate for required text. Spot UV highlights brand elements without crowding the warning panel. Embossing on the brand name or a leaf motif adds tactile depth that communicates premium quality.
Kraft packaging printed with soy-based inks hits hard with the sustainability-conscious CBD buyer. A "100% recyclable" callout on the bottom panel or an FSC certification mark costs nothing extra but signals the environmental values this audience actively shops for.
The layout hierarchy that works best across compliant CBD boxes: brand and product name at the top, potency and serving data in the middle, warnings and regulatory copy at the bottom, QR code on the side panel linking to COA results. Clean, functional, and nothing hidden. That transparency is the brand story.
Build It Right Before the First Print Run
Reworking non-compliant CBD packaging after production is expensive, stressful, and completely avoidable. The brands that invest in getting the design, labeling, and compliance structure right on the first run never have to deal with recalls, reprints, or lost retail relationships.
We build regulation-ready CBD packaging for oils, gummies, pre-rolls, topicals, and vape products. Free compliance-aware design support, no plate fees, free shipping across the USA. MOQ starts at 100 units. Get your free quote and design consultation today.
What CBD Brands Need to Know Before Ordering
On child-resistant certification:
Required for all ingestible CBD products. Packaging must be tested under 16 CFR §1700.20. Ask your supplier for documentation matching your exact container and closure setup.
On label content:
Product name with CBD declaration, mg potency per serving and per package, ingredients, batch number, manufacturer address, net weight, and state-required warnings. No shortcuts.
On health claims:
Prohibited unless FDA-approved. Wellness language works. Medical language triggers enforcement. Get legal review before printing.
On state compliance:
Design for the strictest state in your distribution footprint. Modular label templates save money as you expand into new markets.
On QR codes:
Link to live, batch-specific COA results. Dead links and generic pages create compliance exposure and erode buyer trust.
On design restrictions:
No imagery that appeals to minors. No candy-style graphics, cartoon characters, or bright palettes resembling children's products. This is the most commonly enforced packaging violation in the CBD space.
